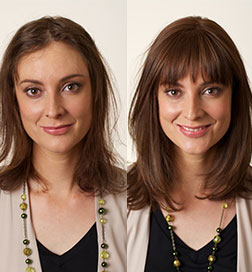This week the Food and Drug Administration (FDA) in the United States approved the first systemic treatment for alopecia areata. Alopecia areata is an auto-immune disease that affects more than 500,000 Australians, and there is currently no cure. It causes the body’s immune system to attack hair follicles in men, women and children, and can result in dramatic hair loss, ranging from bald patches to total baldness.
What is Olumiant?
The medication is known as Olumiant and contains the drug baricitinib. It is available in oral tablets, taken once daily. The FDA has approved Olumiant for adults with severe alopecia. About 40% of people saw significant hair regrowth in two large trials that spanned over 36 weeks.
Olumiant has traditionally been used to treat moderate to severe rheumatoid arthritis. It works by disrupting the communication of specific enzymes involved in harming hair follicles and causing inflammation, and it can help to calm down an overactive immune system. Olumiant isn’t recommended for use with other immunosuppressant drugs. Common side effects of Olumiant can include upper respiratory tract infections, headache, acne, high cholesterol and other medical conditions.
Will Olumiant be available in Australia?
The Therapeutic Goods Association (TGA) is monitoring its use as a treatment for alopecia here in Australia.
This is very exciting news for those who suffer from alopecia areata, as there currently aren’t many medical options for them. This will be the first FDA-approved systemic medicine to treat it, and we know many are hoping the TGA soon follows suit. Watch this space!